About Event
The LNP Characterization & Analytical Development Summit is your leading forum to benchmark with the LNP community to dive deep into the biggest characterization and analytical development challenges facing the field.
Join Global Investors to:
Focus in on the latest strategies to comprehensively characterize structural properties of complex lipid nanoparticles, including both static and dynamic properties

Maximize control through standardized sample preparation and align with process development requirements

Manage the diverse interactions with LNP components and the payload, indicating the endosomal release outcomes

Develop a robust potency assay to reflect the mechanisms of action and total functionality of the LNP

Benchmark analytical requirements across phase development and enlighten optimal cross department communication

Equip the analytical data packages with in-depth immunogenicity, stability and impurity assays to minimize batch variability
This summit was dedicated to this emerging group of scientists to equip them with the quantitative techniques, and leveraging this to support formulation & process development group to enhance product quality.
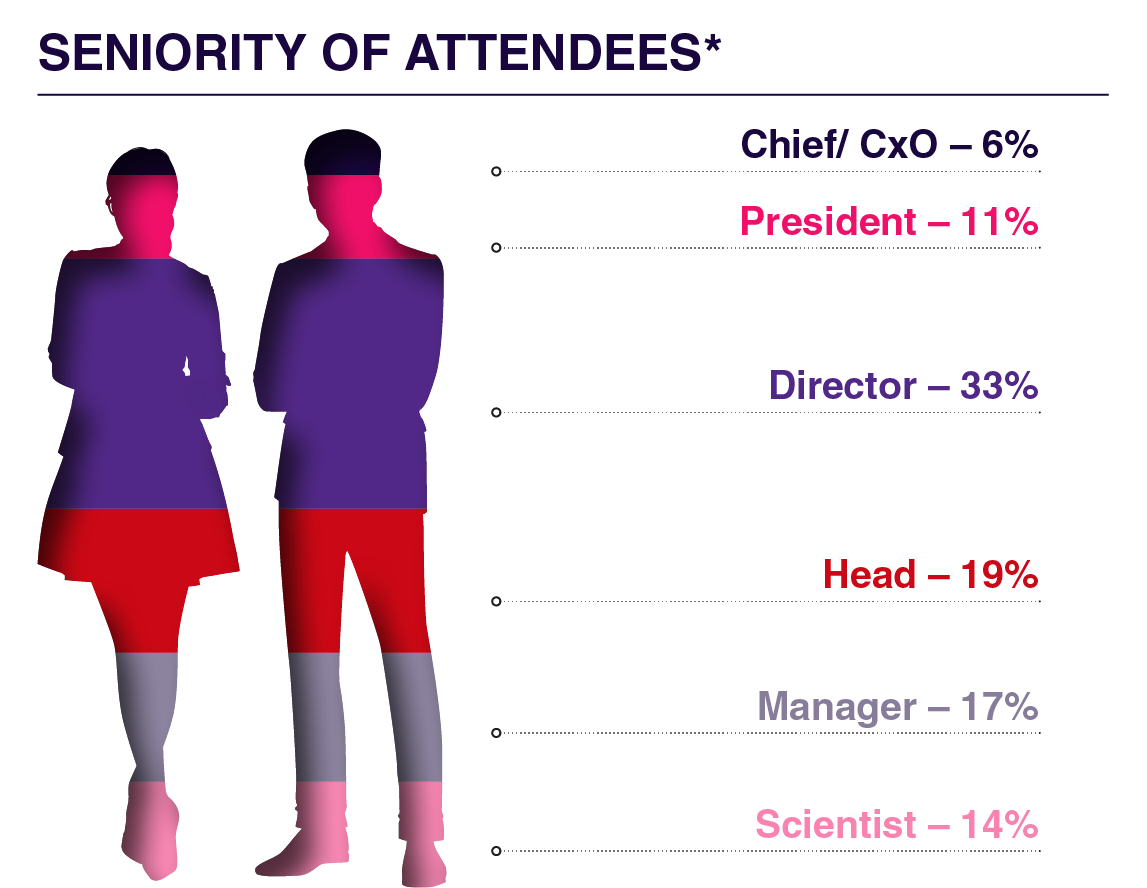
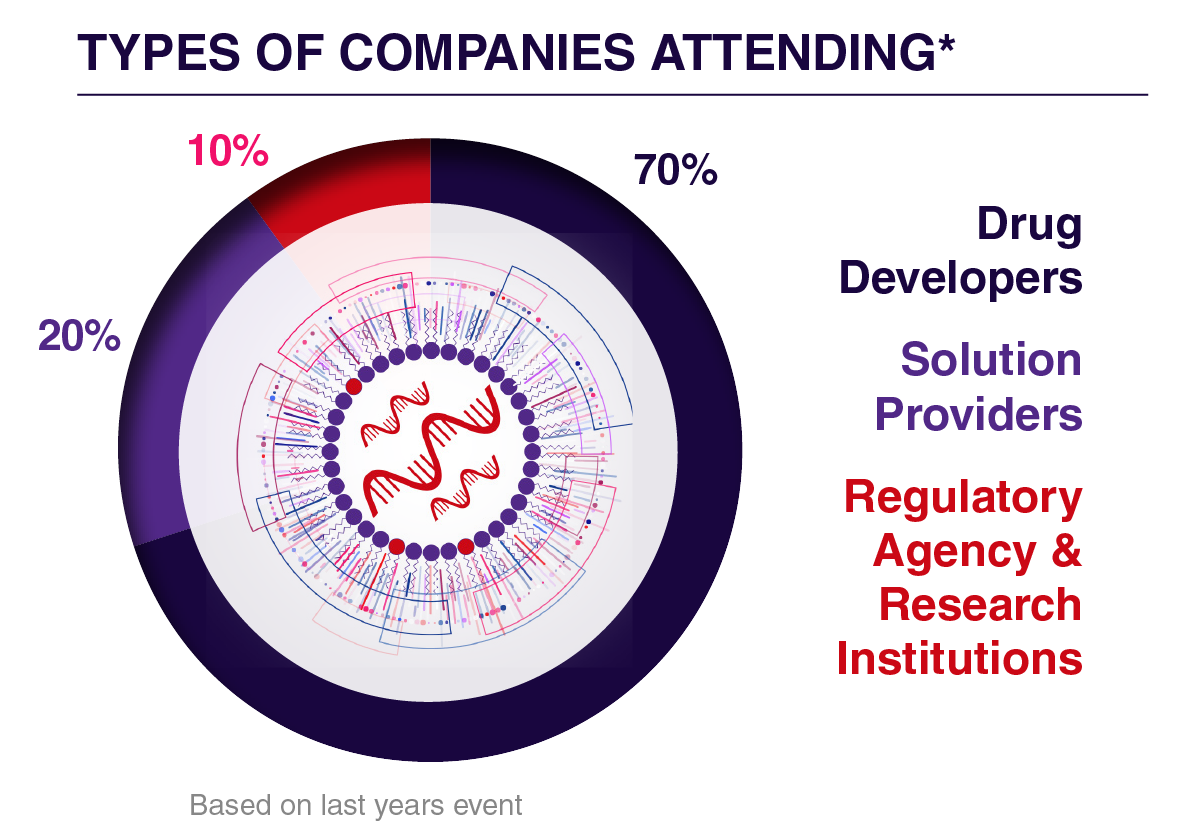
What Previous Attendees of the LNP Series Had To Say:
"Fantastic networking opportunity and learning about latest development in the field from speakers and audience."


